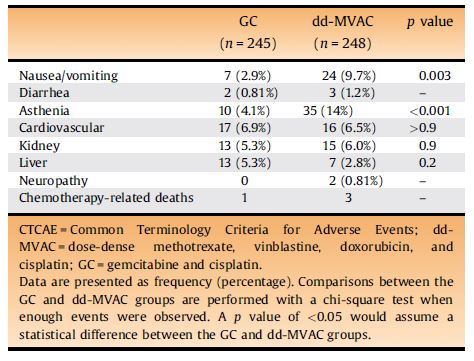
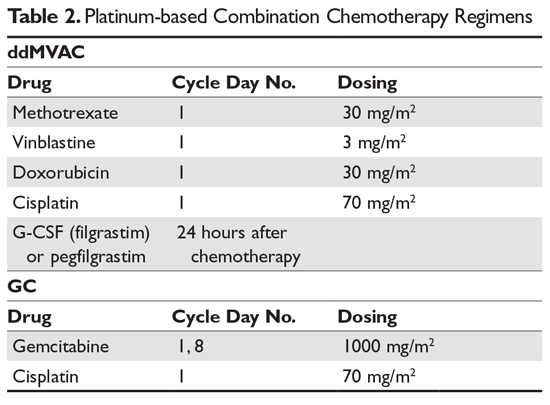
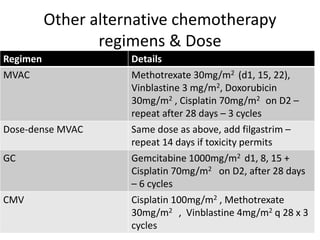
Neoadjuvant Dose-dense Gemcitabine and Cisplatin in Muscle-Invasive Bladder Cancer: Results of a Phase 2 Trial - European Urology Oncology

A practical approach to bladder preservation with hypofractionated radiotherapy for localised muscle-invasive bladder cancer - Clinical and Translational Radiation Oncology

Dose-Dense Methotrexate, Vinblastine, Doxorubicin, and Cisplatin With or Without Panitumumab in Patients With Advanced Urothelial Carcinoma: Multicenter, Randomized, French Unicancer GETUG/AFU 19 Study - Clinical Genitourinary Cancer

Comparison of clinical outcomes in patients with localized or locally advanced urothelial carcinoma treated with neoadjuvant chemotherapy involving gemcitabine–cisplatin and high dose-intensity MVAC | SpringerLink

ESMO 2021: VESPER Trial Indicates Neoadjuvant Dose-Dense MVAC May be Practice-Impacting in Muscle Invasive Bladder Cancer. | PracticeUpdate
Neoadjuvant Dose Dense MVAC versus Gemcitabine and Cisplatin in Patients with cT3-4aN0M0 Bladder Cancer Treated with Radical Cys

Prospective, open-label, randomized, phase III study of two dose-dense regimens MVAC versus gemcitabine/cisplatin in patients with inoperable, metastatic or relapsed urothelial cancer: a Hellenic Cooperative Oncology Group study (HE 16/03) - Annals

Dose-Dense Methotrexate, Vinblastine, Doxorubicin, and Cisplatin or Gemcitabine and Cisplatin as Perioperative Chemotherapy for Patients With Nonmetastatic Muscle-Invasive Bladder Cancer: Results of the GETUG-AFU V05 VESPER Trial | Journal of Clinical ...

EAU15 - Poster: Neoadjuvant dose-dense MVAC for muscle invasive bladder cancer; efficacy and safety compared to the traditional MVAC regimen

GETUG on Twitter: "VESPER (NCT01812369) phase III trial in bladder cancer comparing cisplatin-gemcitabine and dose-dense MVAC in perioperative setting. Preliminary results at ASCO GU 2020 https://t.co/kpjFOQLmap" / Twitter

Dose-dense MVAC vs gemcitabine and cisplatin tied to better survival in bladder cancer patients | Latest news for Doctors, Nurses and Pharmacists | Pharmacy

1438-Bladder/Urothelial neoadjuvant MVAC (methotrexate vinBLASTine DOXOrubicin ciSplatin) SUPERSEDED | eviQ

Randomized Phase III Trial of Dose-dense Methotrexate, Vinblastine, Doxorubicin, and Cisplatin, or Gemcitabine and Cisplatin as Perioperative Chemotherapy for Patients with Muscle-invasive Bladder Cancer. Analysis of the GETUG/AFU V05 VESPER Trial ...
MVAC Chemotherapy Toxicity - Activity Seven:<br>Role of Neoadjuvant Chemotherapy in Bladder Cancer - Educational Centre:<br>Current Clinical Problems in Bladder Cancer

VESPER: Phase III Trial of Perioperative Chemotherapy With Dose-Dense MVAC vs Gemcitabine + Cisplatin in Patients With Muscle-Invasive Bladder Cancer
Neoadjuvant chemotherapy with dose dense MVAC is associated with improved survival after radical cystectomy compared to other cytotoxic regimens: A tertiary center experience | PLOS ONE

Chemotherapy for Muscle-invasive Bladder Cancer: Impact of Cisplatin Delivery on Renal Function and Local Control Rate in the Randomized Phase III VESPER (GETUG-AFU V05) Trial - Clinical Genitourinary Cancer
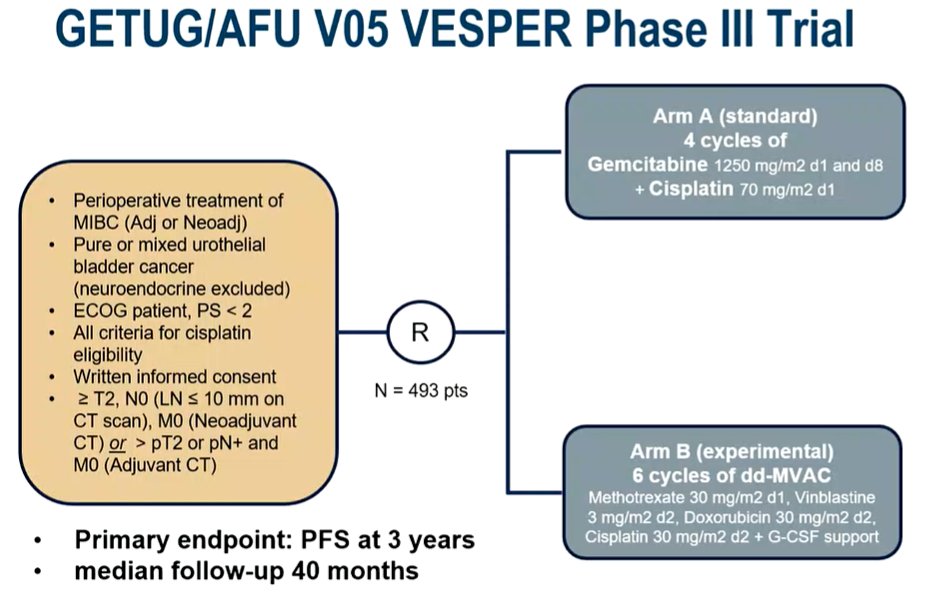
ESMO 2021: GETUG/AFU VESPER V05 Phase III Trial of Dose Dense Methotrexate, Vinblastine, Doxorubicin and Cisplatin or Gemcitabine and Cisplatin As Perioperative Chemotherapy for Patients With MIBC

PD11-10 - Neoadjuvant Dose-Dense MVAC increases down-staging and improves survival compared to other chemotherapy regimens