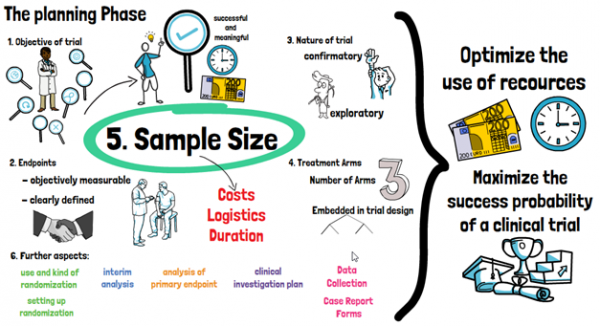
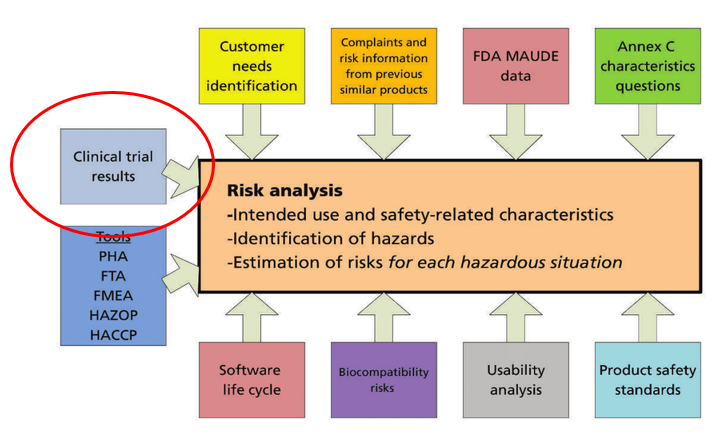
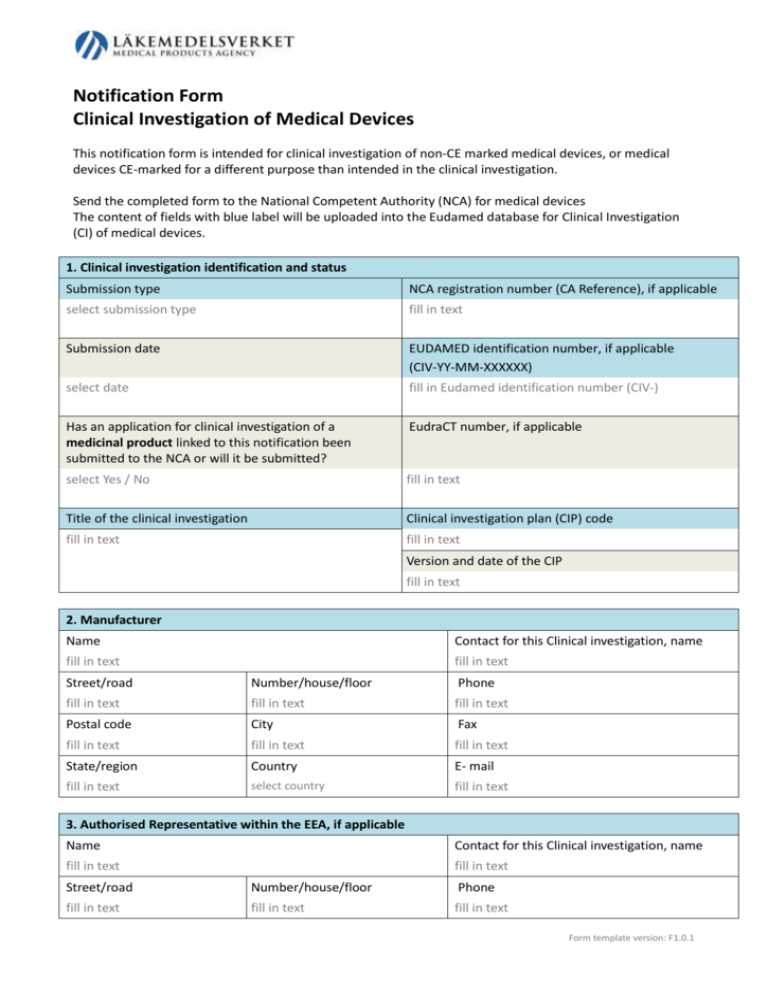
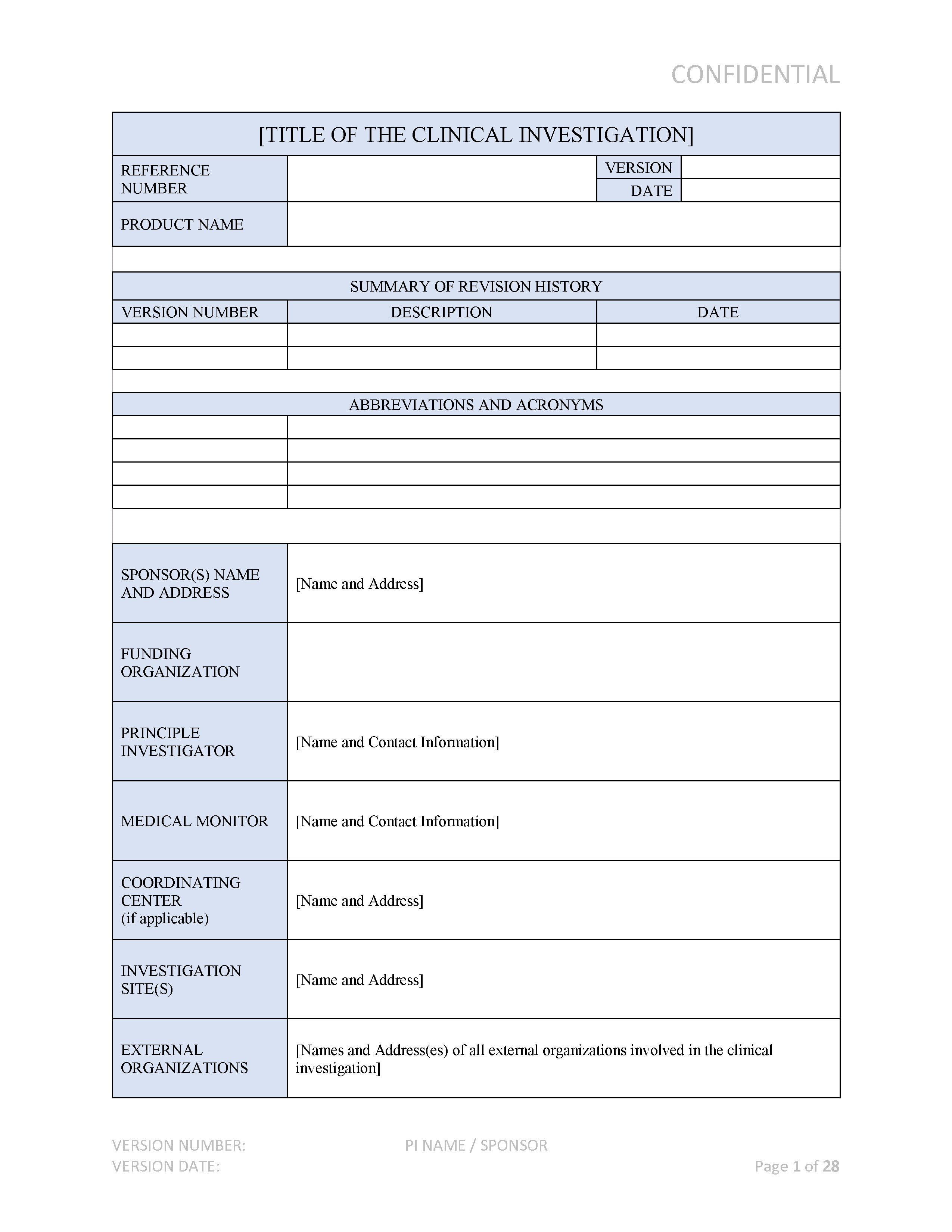
GUIDELINES ON MEDICAL DEVICES GUIDELINES ON CLINICAL INVESTIGATION: A GUIDE FOR MANUFACTURERS AND NOTIFIED BODIES
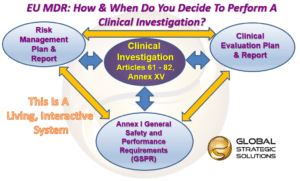
What Is The Difference Between Clinical Evaluation and Clinical Investigation? | Global Strategic Solutions

What you Need and When – The Key Documents in the Drug Lifecycle - Trilogy Writing & Consulting GmbH

PDF) Clinical investigation plan for the use of interactive binocular treatment (I-BiT) for the management of anisometropic, strabismic and mixed amblyopia in children aged 3.5–12 years: a randomised controlled trial