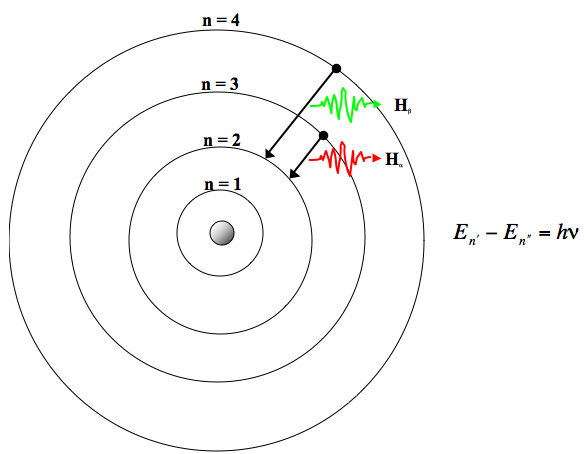
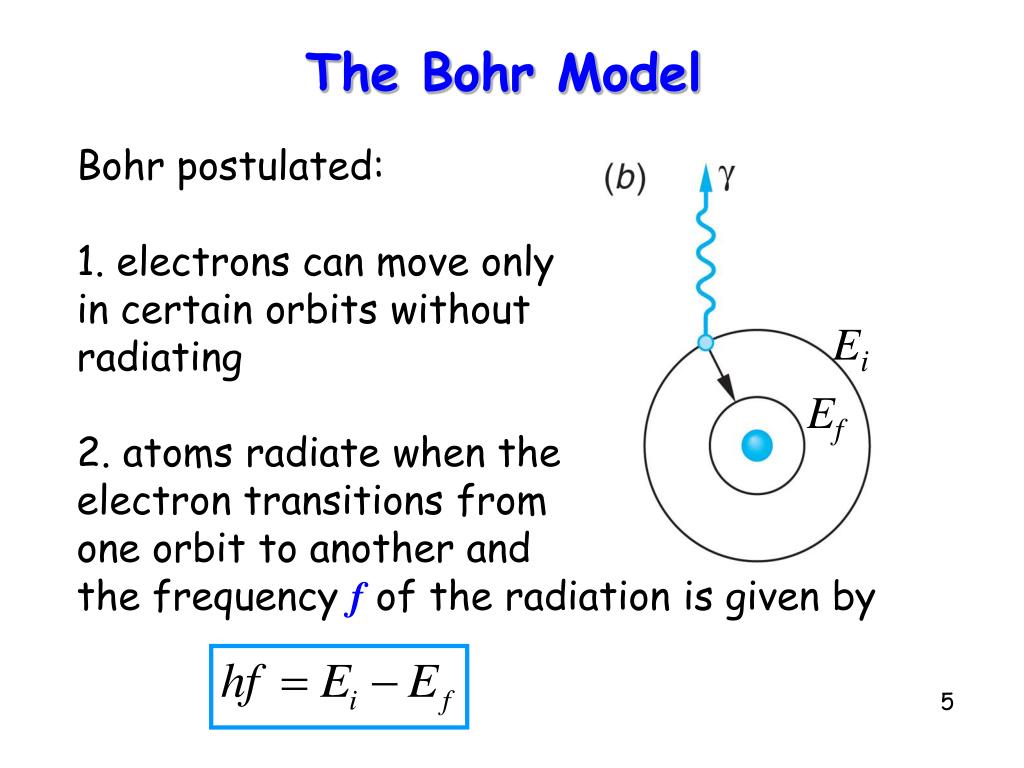
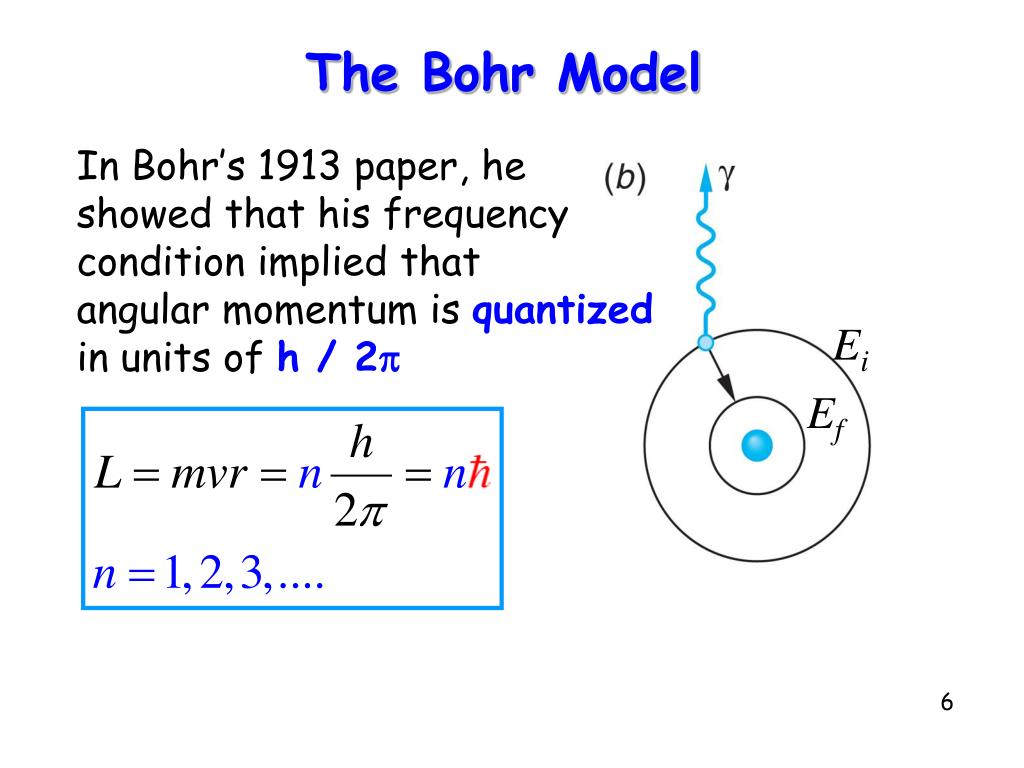
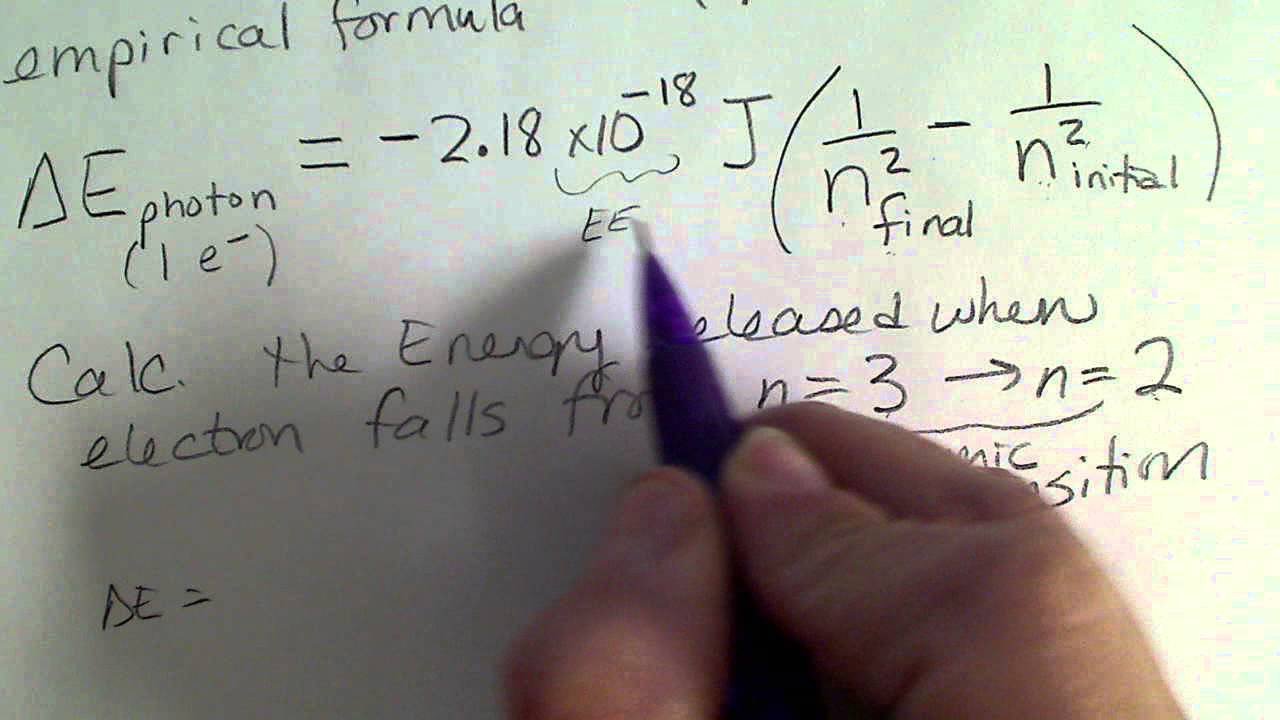Using Bohr's postulates, derive the expression for the frequency of radiation emitted - Sarthaks eConnect | Largest Online Education Community

Calculate the angular frequency of an electron occuppying the second Bohr orbit of `He^(+)` ion. - YouTube
In the Bohr atom model, the frequency of transitions is given by the following expression - Sarthaks eConnect | Largest Online Education Community
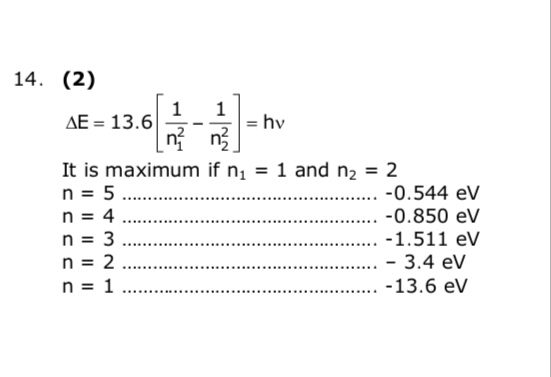
According to Bohr atom model, in which of the following transitions will the frequency be maximum ? - Sahay Sir

Calculate frequency of revolution of electron in 4th Bohr orbit `Be^(+3)` ion . Given that `(pi^(2 - YouTube
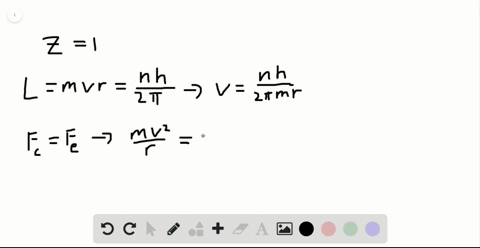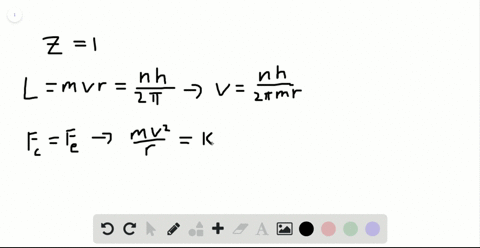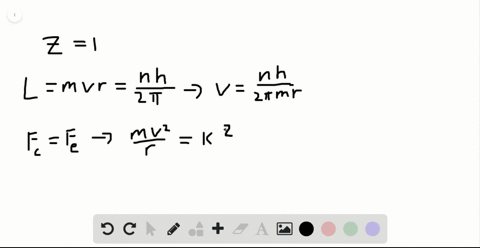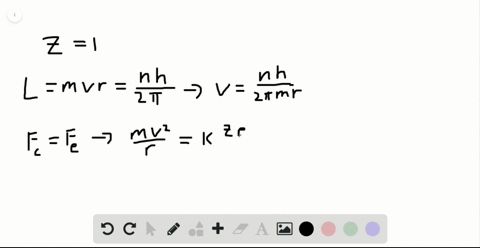
SOLVED:Determine the speed and frequency of an electron moving around the first Bohr orbit in hydrogen. According to classical physics, the atom should emit electromagnetic radiation at this frequency. In what portion
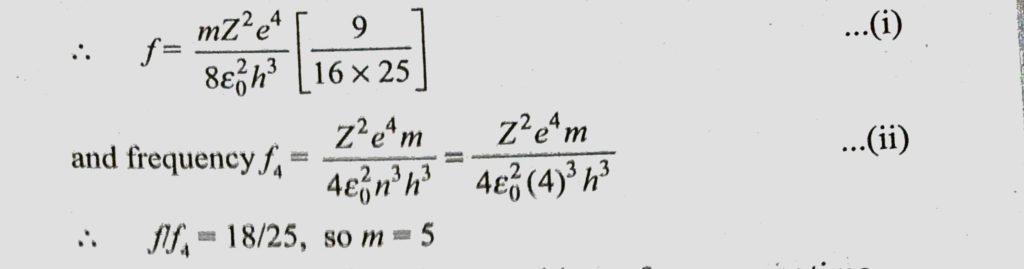
A Bohr hydrogen atom undergoes a transition n = 5 to n = 4 and emits a photon of frequency f. Frequency of circular motion of electron in n = 4 orbit

What is the frequency of revolution of electron present in `2nd` Bohr\'s orbit of `H-` atom ? - YouTube

Calculate frequency of revolution of electrons in `4^(th)` Bohr orbit of `Be^(+3)` ion. Given that - YouTube