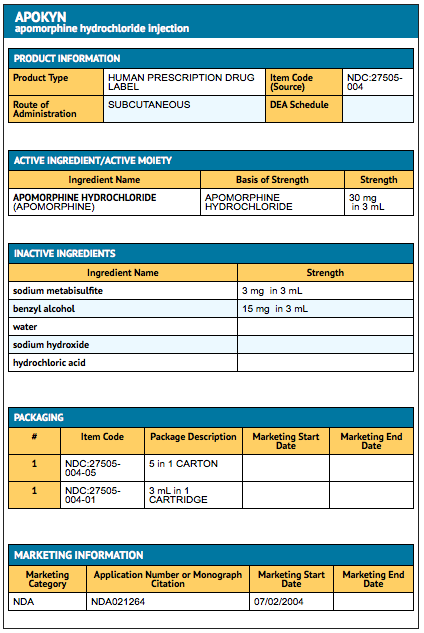
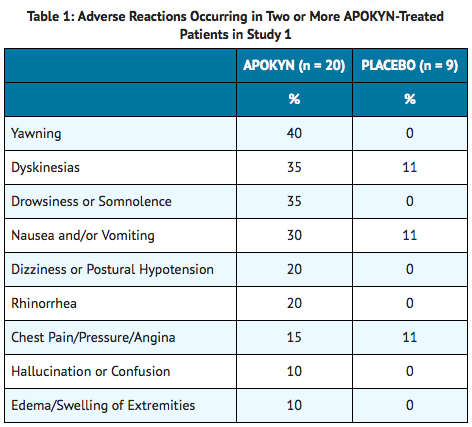
Apomorphine subcutaneous infusion reduces dopaminergic dosage whilst maintaining motor benefits in Parkinson's disease: A prospective analysis of Thai Apomorphine Registry - MDS Abstracts

A case series of rapid titration of subcutaneous apomorphine in Parkinson's disease | British Journal of Neuroscience Nursing
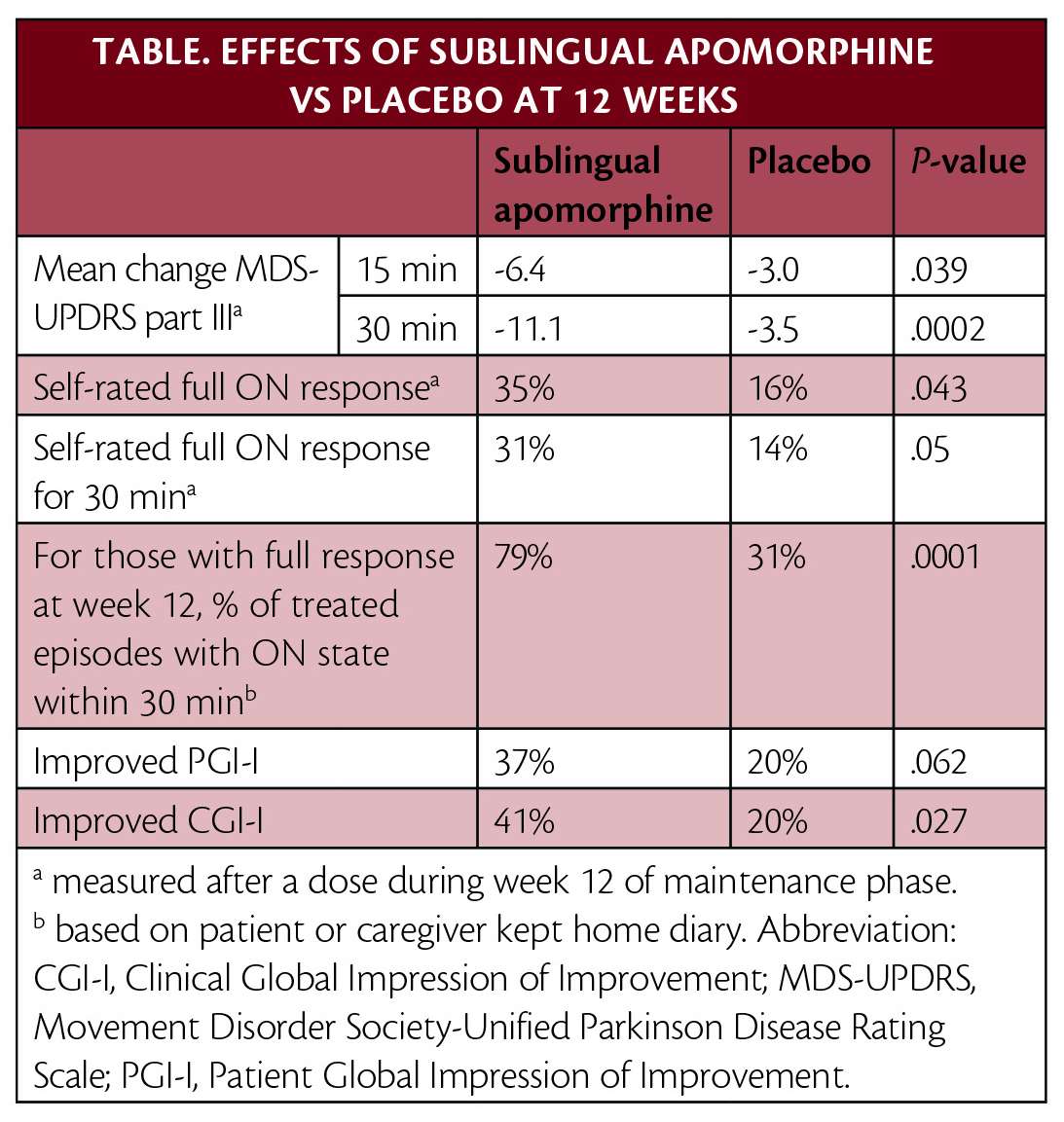
FDA Approves Apomorphine Sublingual Film for Treatment of OFF Periods in Parkinson Disease - Practical Neurology

Sunovion Pharmaceuticals Inc. - Sunovion Announces U.S. FDA Approval of KYNMOBI™ (apomorphine hydrochloride) Sublingual Film for the Treatment of Parkinson's Disease OFF Episodes
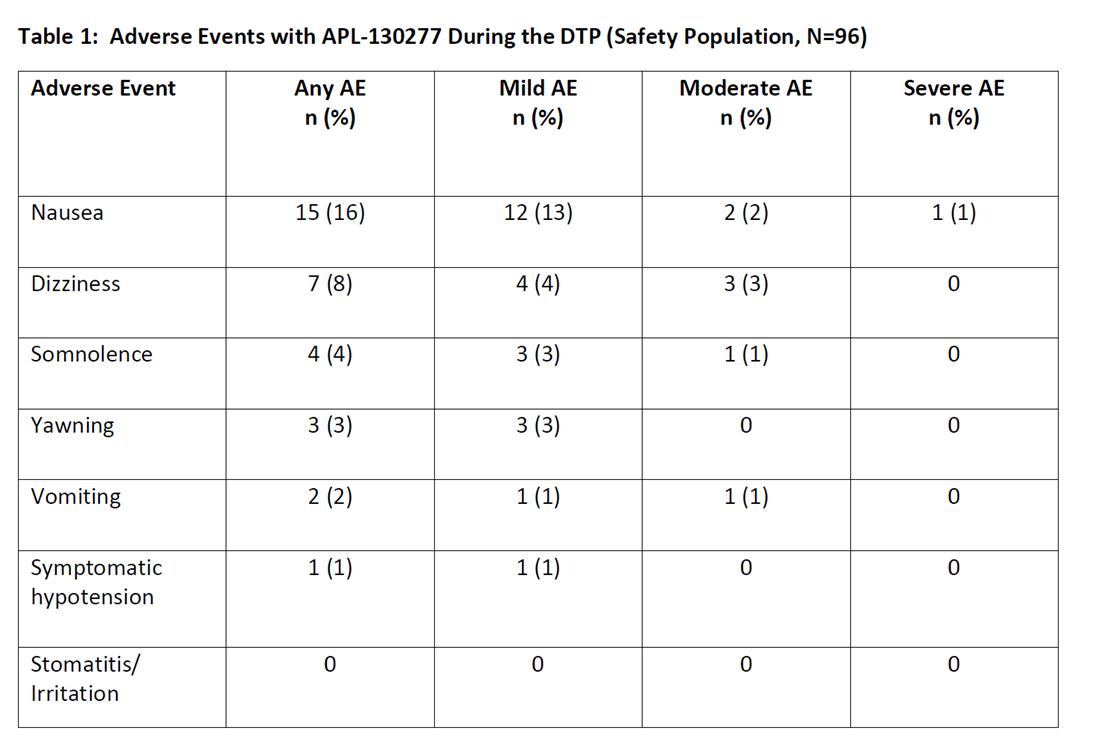
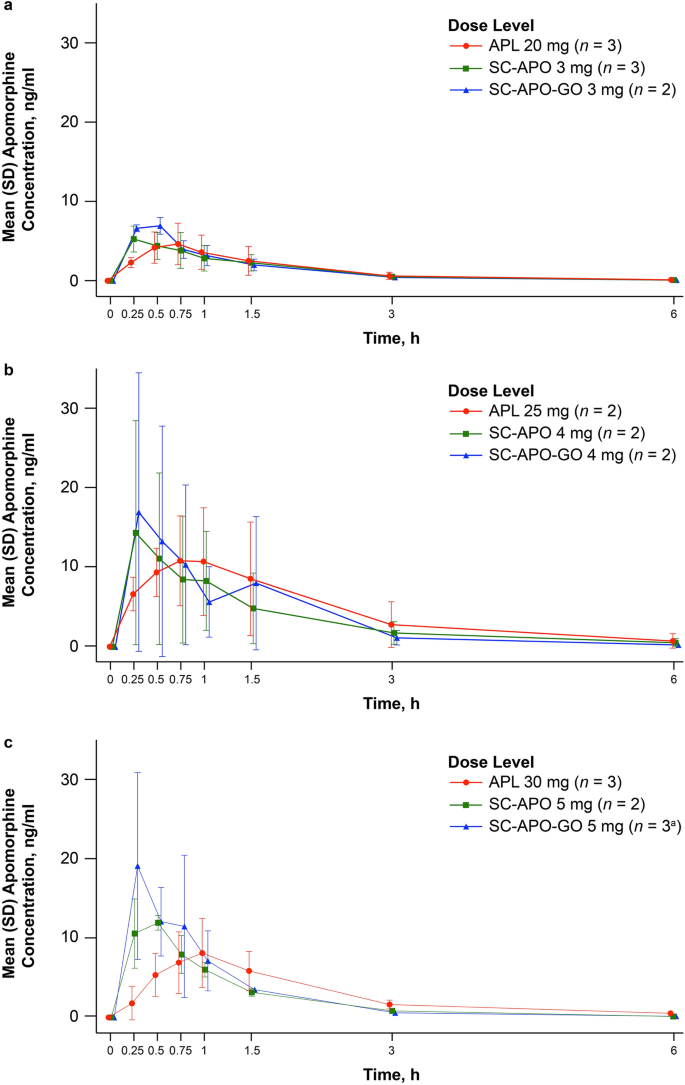
Safety of sublingual apomorphine film (APL-130277) for the treatment of OFF episodes in patients with Parkinson's disease: Preliminary Results from a Phase III Study - MDS Abstracts

Apomorphine subcutaneous infusion reduces dopaminergic dosage whilst maintaining motor benefits in Parkinson's disease: A prospective analysis of Thai Apomorphine Registry - MDS Abstracts

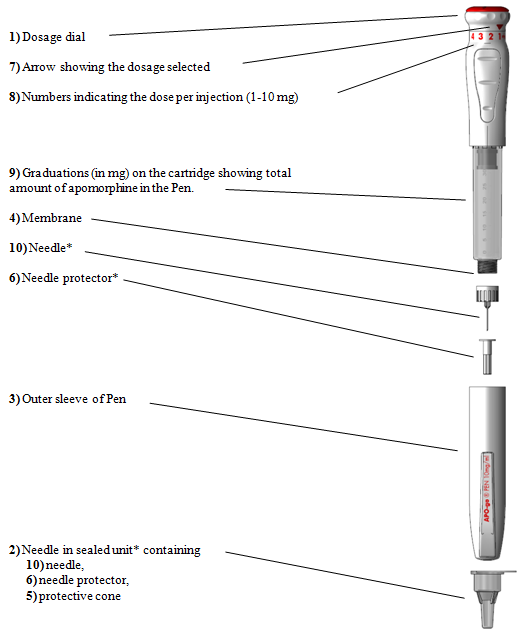
Apomorphine subcutaneous infusion in patients with Parkinson's disease with persistent motor fluctuations (TOLEDO): a multicentre, double-blind, randomised, placebo-controlled trial - The Lancet Neurology

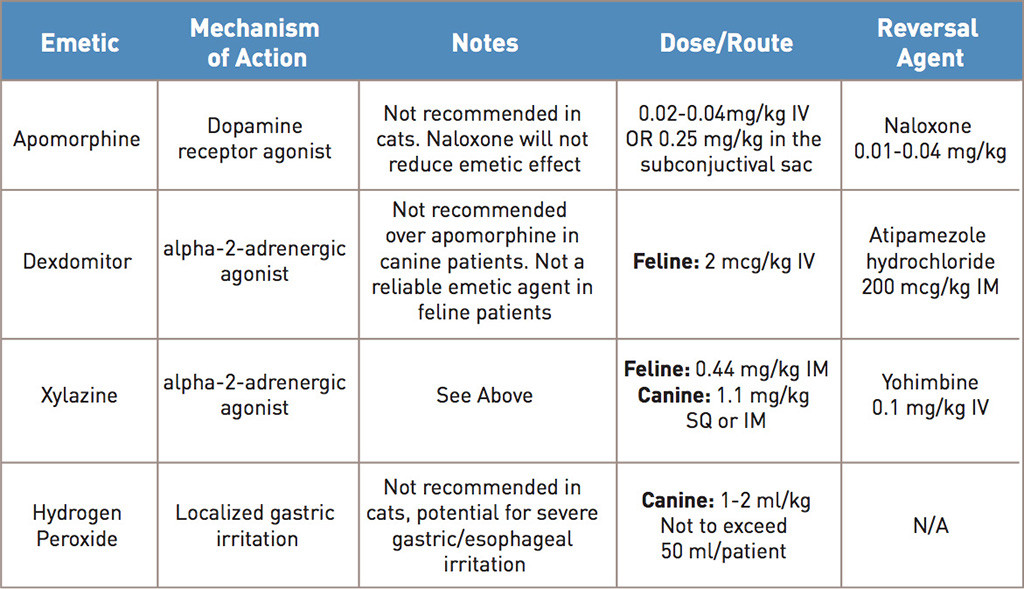
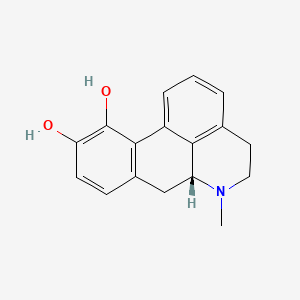
PDF) Challenges and trends in apomorphine drug delivery systems for the treatment of Parkinson's disease
Sunovion Announces U.S. FDA Approval of KYNMOBI™ (apomorphine hydrochloride) Sublingual Film for the Treatment of Parkinson's Disease OFF Episodes | Business Wire