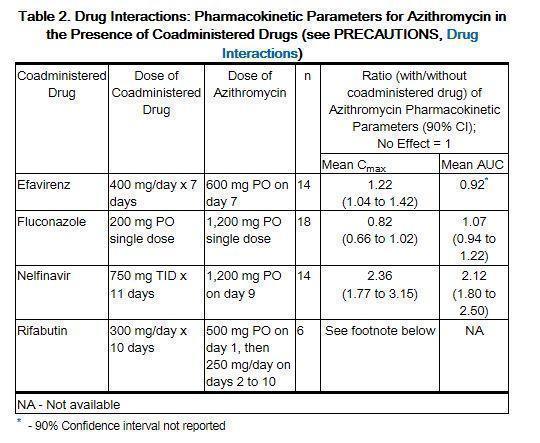
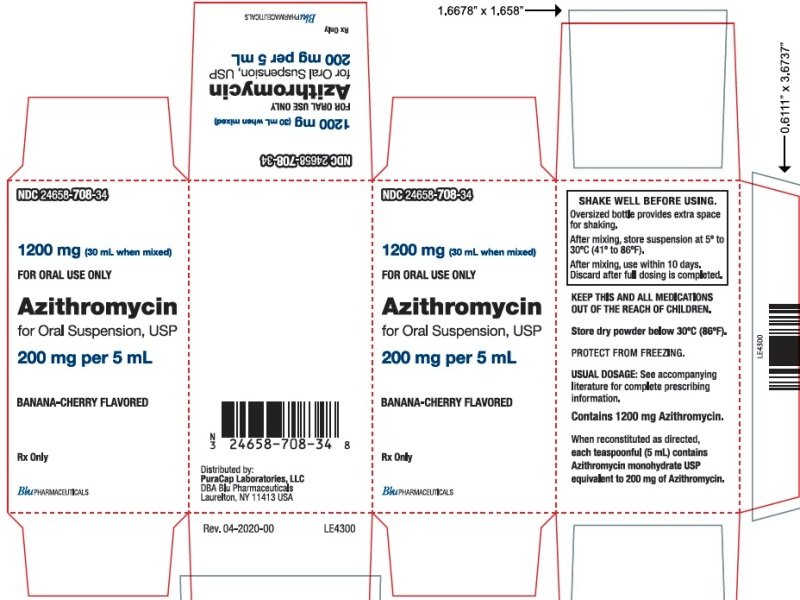
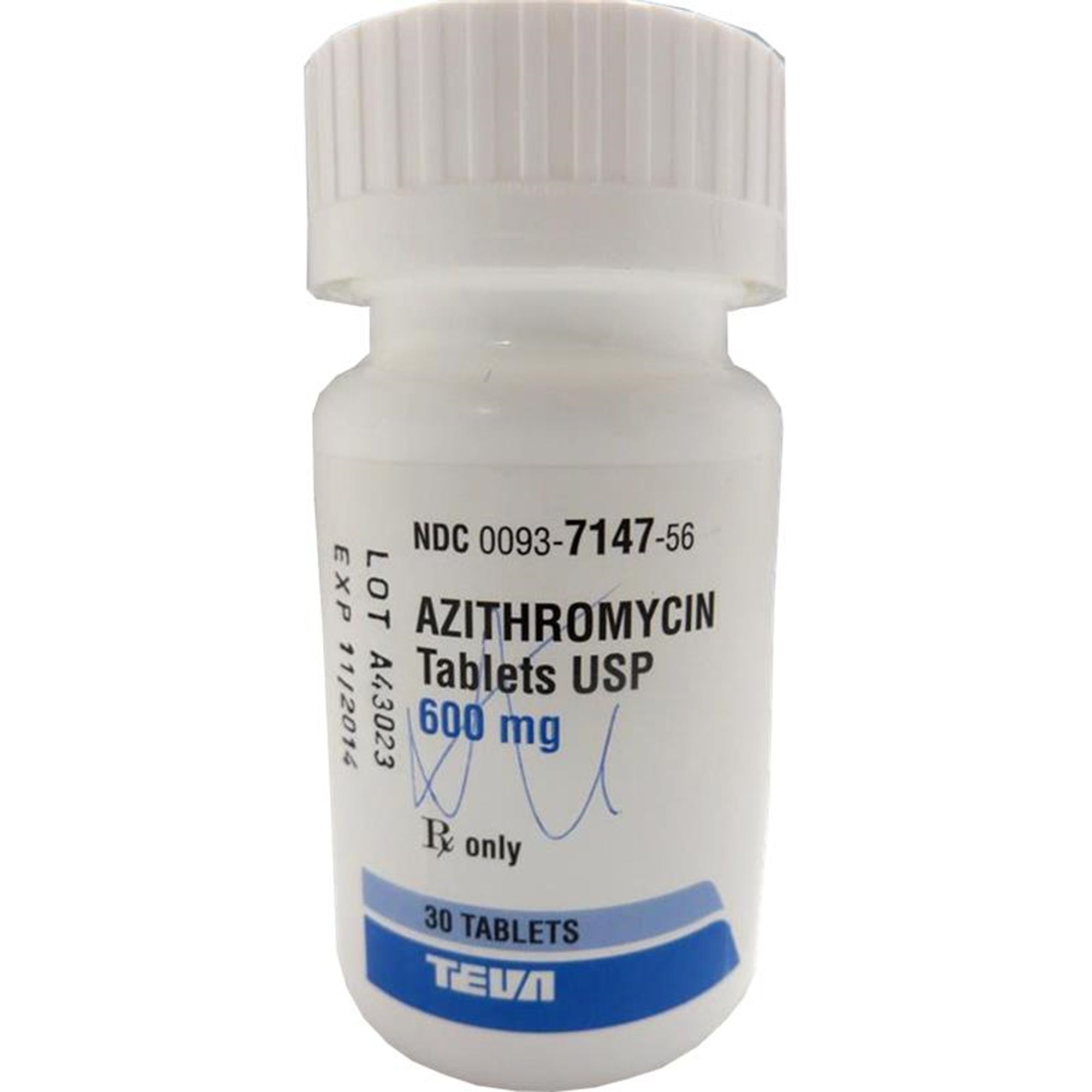
These highlights do not include all the information needed to use AZITHROMYCIN TABLETS safely and effectively. See full prescribing information for AZITHROMYCIN TABLETS. AZITHROMYCIN tablets, for oral use Initial U.S. Approval: 1991

Comparative pharmacokinetics of azithromycin in serum and white blood cells of healthy subjects receiving a single-dose extended-release regimen versus a 3-day immediate-release regimen. - Abstract - Europe PMC

A Reference‐Scaled Average Bioequivalence Study of Azithromycin Tablets Manufactured in China and the United States: An Open‐Label, Randomized, Single‐Dose, 3‐Way Crossover Study in Healthy Chinese Subjects Under Fasted and Fed Conditions -

Azithromycin for community treatment of suspected COVID-19 in people at increased risk of an adverse clinical course in the UK (PRINCIPLE): a randomised, controlled, open-label, adaptive platform trial - The Lancet